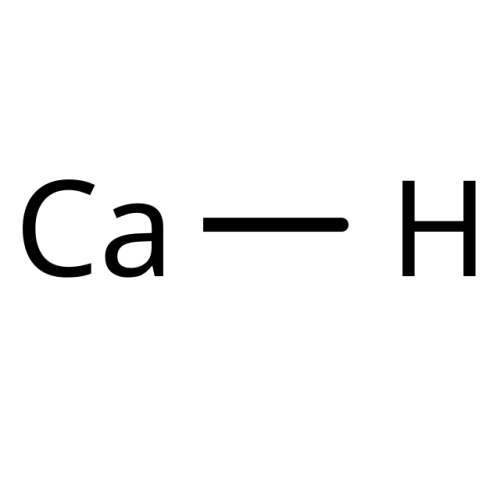
Calcium Hydride, Grade S
CaH2
Calcium Hydride, Grade S (CAS 7789-78-8, molecular formula CaH2) is an inorganic chemical supplied as a technical-grade material. It is typically a white to grey powder.
Calcium hydride is used primarily as a source of hydrogen, as a drying agent for liquids and gases, and as a reducing agent for metal oxides.
Typical specifications include: Ca total min. 92 %; H min. 980 ml/g CaH2; Mg max. 0.8 %.
Flammable solid. Contact with water liberates highly flammable gases!
Applications
Calcium hydride is used primarily as a source of hydrogen, as a drying agent for liquids and gases, and as a reducing agent for metal oxides.
Specifications
- Ca total min. 92 %
- H min. 980 ml/g CaH2
- Mg max. 0.8 %
- N max. 0.2 %
- Al max. 0.01 %
- Cl max. 0.5 %
- Fe max. 0.01 %
Method of Analysis
Calcium complexometric, impurities by spectral analysis and special analytical procedures. Gas volumetric determination of hydrogen. Produces with water approx. 1,010 ml hydrogen per gram.Handling Instructions
Flammable solid. Contact with water liberates highly flammable gases! Calcium hydride decomposes partially and reversibly at temperatures above 600 °C. Calcium hydride is insoluble in most organic solvents. Upon direct contact with water Calcium hydroxide [Ca(OH)2] and pure hydrogen gas are produced in a violent reaction and self-ignition is possible. One kg of CaH2 liberates approx. 1 m3 of hydrogen. Avoid contact with water and with skin. Wear protective goggles and gloves and avoid formation of dust. In case of fire cover with dry sand, calcined soda or quicklime. Never use water, carbon dioxide, or halocarbon extinguisher. Should be handled with minimal exposure to humid air.
Storage Instructions
Store cool and dry in airtight containers away from heat sources.
Legal Disclaimer
The information presented herein is believed to be accurate and reliable but is presented without guarantee or responsibility on the part of Albemarle Corporation and its subsidiaries and affiliates. It is the responsibility of the user to comply with all applicable laws and regulations and to provide for a safe workplace. The user should consider any health or safety hazards or information contained herein only as a guide and should take those precautions which are necessary or prudent to instruct employees and to develop work practice procedures in order to promote a safe work environment. Further, nothing contained herein shall be taken as an inducement or recommendation to manufacture or use any of the herein materials or processes in violation of existing or future patent.